Density
Core Concept
Density is the amount of mass per given volume. It is expressed as density = mass/ volume
Think of density as “how much stuff is in a given space.” Usually the space is considered a 1 cubic block, for comparisons.
Master the Formula: Understand and apply Density=Mass/Volume, ensuring units are consistent.
Practice Unit Conversions: Convert between mass and volume units, like g to kg or $\text{cm}^3$ to mL.
Sink or Float: Calculate density for objects and predict if they will sink or float based on water's density.
Technique to measure volume: Use tools like water displacement for irregular volumes and lab equipment for measurements.
Solve Problems and Graph: Rearrange the formula to solve for mass or volume and graph mass vs. volume to find density.
Test Yourself
Podcast Episode
Practice Problems & Worked Out Examples 🔒
Browse by category
Direct Calculation and Variable Manipulation
→ 02Density as an Identifying Intensive Property
→ 03Geometric and Displacement Volume Problems
→ 04Layering, Buoyancy, and Relative Density
→ 05Unit Conversions and Complex Units
→ 06Temperature and Phase Effects on Density
M
→ 07Other / Uncategorized
→ 08Assorted Multiple Choice
→Definition
Common units for density include: grams/ milliliter (g/mL) or grams/cubic centimeter (g/cm3)
Density is an intensive property meaning that it is value specific to the substance, no matter amount. If you have a huge block of copper or just a small cube -- the density of both will always be the same, since it is copper. For this reason, if you have an unknown substance you can find its density and try to match it to a list of known substances and their density to help identify it.
Calculating Density with a Graph:
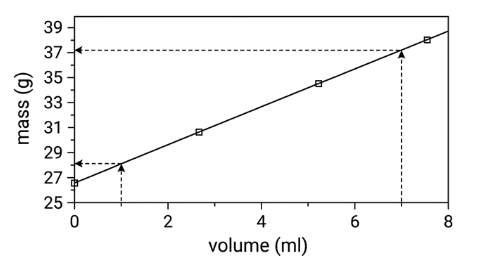
Density on graph: y-axis is mass; x-axis is volume.
Therefore the density = slope of the line.
Slope = Δy/ Δx = mass / volume (= the formula for density)
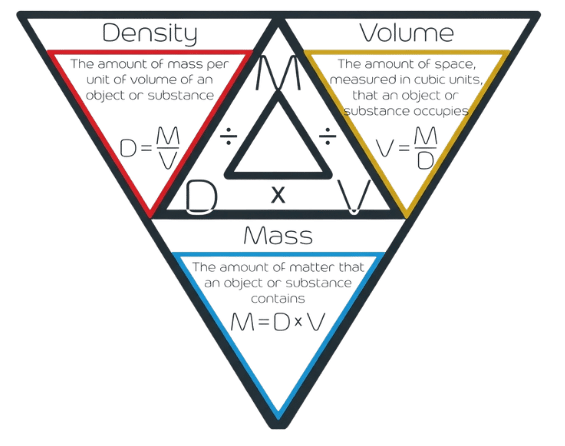
🧠 How to Use the Density Triangle 🧠
The density triangle is a memory aid used to rearrange the formula. By visualizing the formula in a triangle, you can easily derive all three variations:
To find Mass, cover m (Multiply). $m = D \times V$
To find Density, cover D (Divide). $D = m/V$
To find Volume, cover V (Divide). $V = m/D$
(Think: "I'm looking for Volume," so I cover the V, and I see m over D.)
Visualize the Triangle: Keep the "triangle structure" in your mind:
Mass is always on top (think of it as the "Master").
Density and Volume are roommates on the bottom.
Topic Related Resources
Lab
Coming Soon...
Lab
Coming Soon...
Demo
Coming Soon...
Demo
Coming Soon...
Activity
Coming soon...
Activity
Coming soon...
Simulation
Coming soon...
Simulation
Coming soon...


