Equilibrium
Core Concept
Equilibrium is a state in a reversible chemical reaction where the forward and reverse reaction rates are equal, resulting in constant concentrations of reactants and products.
The reaction is dynamic—reactants and products continue to interconvert, but their concentrations remain unchanged.
Equilibrium can be achieved from either direction (starting with reactants or products).
Equilibrium expressions quantitatively describe the balance between reactants and products in a reversible chemical reaction at equilibrium, using the ratio of their concentrations or partial pressures raised to the power of their stoichiometric coefficients.
The equilibrium expression relates the concentrations or partial pressures of reactants and products at equilibrium using the reaction's stoichiometry.
For a reaction aA + bB ↔ cC + dD, the equilibrium constant (K) is written as $K = \frac{[C]^c[D]^d}{[A]^a[B]^b}$.
Only gases and aqueous species are included in the equilibrium expression; solids and liquids are excluded.
The value of K indicates the reaction's favorability; K>1 favors products, while K<1 favors reactants.
The equilibrium constant Kc uses molar concentrations, and $K_p$ uses partial pressures for gaseous reactions.
$K_p$ and $K_c$ are related by $K_p = K_c(RT)^{\Delta n}$, where Δn is the change in moles of gas.
Test Yourself
Podcast Episode
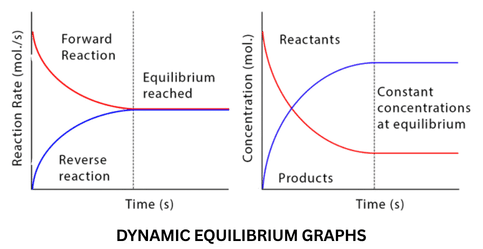
Chemical equilibrium is the state in which the rate of the forward reaction equals the rate of the reverse reaction, resulting in no net change in the concentrations of reactants and products.
The Nature of Dynamic Equilibrium
Reversibility: In a closed system, most reactions can proceed in both directions. We use the double arrow ($\rightleftharpoons$) to indicate this.
The "Dynamic" Aspect: At equilibrium, the macroscopic properties (color, pressure, concentration) stay constant, but at the molecular level, bonds are constantly breaking and forming. The forward and backward "traffic" is moving at the exact same speed.
Closed Systems: Equilibrium can only be established in a closed system where no matter can escape. If a gas product escapes, the reaction will never reach equilibrium.
Chemical Equilibrium Basics
Definition: Reactions that can proceed in both forward and reverse directions simultaneously.
Notation: The use of the double arrow ($\rightleftharpoons$) indicates that the system is reversible.
Reaction Components
Full System: $\text{A} + \text{B} \rightleftharpoons \text{C} + \text{D}$
Forward Reaction: $\text{A} + \text{B} \rightarrow \text{C} + \text{D}$
Reverse Reaction: $\text{C} + \text{D} \rightarrow \text{A} + \text{B}$
The Equilibrium Constant ($K_{eq}$)
The Law of Mass Action: For any balanced equation $aA + bB \rightleftharpoons cC + dD$, the ratio of products to reactants is constant at a specific temperature.
$$K_{eq} = \frac{[C]^c [D]^d}{[A]^a [B]^b}$$
Pure Solids and Liquids: We exclude pure solids ($s$) and pure liquids ($l$) from the expression because their density (concentration) does not change significantly during the reaction. We only include gases ($g$) and aqueous solutions ($aq$).
Interpreting the Magnitude of $K$:
$K \gg 1$: The equilibrium "lies to the right," meaning the mixture is mostly products.
$K \ll 1$: The equilibrium "lies to the left," meaning the mixture is mostly reactants.
$K \approx 1$: There are significant amounts of both reactants and products.
The Equilibrium Constant (K)
$K_c$ (Concentration Constant): Calculated using the molar concentrations (molarity, $M$) of solutes in a solution or gases in a mixture.
$K_p$ (Pressure Constant): Calculated using the partial pressures of gases (typically in $atm$ or $torr$). This is only applicable to systems containing gases.
Key Formulas
For the general reaction $a\text{A} + b\text{B} \rightleftharpoons c\text{C} + d\text{D}$:
For $K_c$:
$$K_c = \frac{[\text{C}]^c [\text{D}]^d}{[\text{A}]^a [\text{B}]^b}$$
For $K_p$:
$$K_p = \frac{(P_{\text{C}})^c (P_{\text{D}})^d}{(P_{\text{A}})^a (P_{\text{B}})^b}$$
