Heat Flow
Core Concept
Heat flow (also called heat transfer) refers to the movement of thermal energy from a region of higher temperature to a region of lower temperature until thermal equilibrium (equal temperature) is reached.
Understand heat flow basics: Heat flows from regions of higher temperature to lower temperature until thermal equilibrium is reached.
Differentiate heat and temperature: Heat is energy transfer due to temperature difference, while temperature is a measure of the kinetic energy of particles.
Identify heat flow methods: Heat can transfer through conduction (direct contact), convection (fluid movement), or radiation (electromagnetic waves).
Test Yourself
Podcast Episode
Practice Problems & Worked Out Examples 🔒
Topic Overview Podcast
Topic Related Resources
|
LABORATORY
|
DEMONSTRATIONS
|
ACTIVITIES
|
VIRTUAL SIMULATIONS
|
Heat Flow Methods
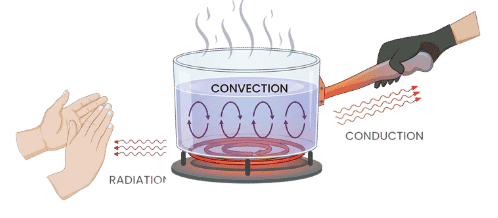
| Feature / Property | Radiation | Convection | Conduction |
|---|---|---|---|
| What it is | Heat transfer through electromagnetic waves; does not require a medium. | Heat transfer through the bulk movement of fluids (liquids or gases) caused by density differences. | Heat transfer through direct contact between molecules or atoms without movement of the substance itself. |
| Medium Required | None (can occur in a vacuum) | Liquids, gases | Solids, liquids, gases |
| Particle Movement | No (electromagnetic waves) | Yes (bulk fluid movement) | No (energy transfer only) |
| How it works | All objects emit thermal radiation based on temperature. Hotter objects emit more at shorter wavelengths. | Heated fluid becomes less dense and rises; cooler, denser fluid sinks, creating a convection current. | Particles vibrate and transfer kinetic energy to neighbors. Metals are efficient due to free-moving electrons. |
| Speed | Fastest (speed of light in vacuum) | Fast (depends on fluid) | Moderate |
| Examples |
|
|
|
| Key Factors |
|
|
|
