Shielding/ Zeff
Core Concept
Definition: The net positive charge experienced by an electron in a multi-electron atom.
Purpose: Reflects the balance between the attraction of electrons to the nucleus and the repulsion caused by other electrons (electron shielding).
Zeff describes the net pull of the nucleus on valence electrons, accounting for electron shielding.
Zeff increases across a period due to increasing proton count and minimal shielding change.
Zeff explains many periodic trends, including atomic radius, ionization energy, and electronegativity.
Test Yourself
Podcast Episode
Practice Problems & Worked Out Examples 🔒
Key Formula
$Z_{\text{eff}}$ = Z - S
Where:
Z: Atomic number (total protons in the nucleus).
S: Shielding constant (a measure of the repulsion by inner electrons).
How $Z_{\text{eff}}$ Works
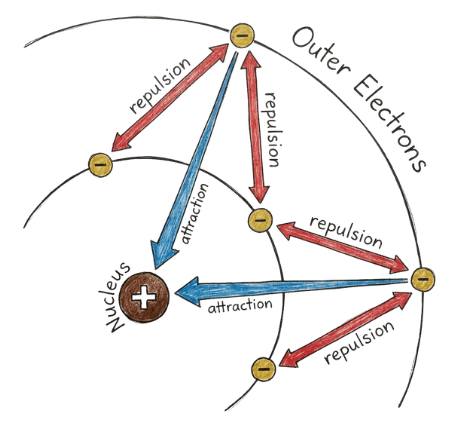
Nucleus Attraction:
Protons in the nucleus attract electrons with a force proportional to Z (the number of protons).
Electron Shielding:
Core (inner) electrons repel valence (outer) electrons, reducing the net positive charge experienced by the valence electrons.
Net Effect:
Valence electrons experience a weaker pull due to shielding but still feel an effective nuclear charge.
Trends in $Z_{\text{eff}}$
Across a Period (Left to Right):
Increases: Protons are added to the nucleus (Z increases), while shielding remains relatively constant.
Result: Electrons are pulled closer to the nucleus, decreasing atomic radius.
Down a Group:
Relatively Constant: Although Z increases, the number of inner electron shells increases significantly, increasing S, so the net Zeff felt by valence electrons changes little.
Applications of $Z_{\text{eff}}$
Atomic Radius: Higher Zeff pulls electrons closer, reducing atomic size across a period.
Ionization Energy: Higher Zeff makes it harder to remove an electron, increasing ionization energy.
Electronegativity: Elements with high Zeff attract bonding electrons more strongly.
Electron Affinity: Higher Zeff results in a more negative electron affinity, as atoms more readily accept electrons.
Topic Related Resources
Lab
Coming Soon...
Lab
Coming Soon...
Demo
Coming Soon...
Demo
Coming Soon...
Activity
Exploring Effective Nuclear Charge: Manipulative Activity
Activity
Coming soon...
Simulation
Effective Nuclear Charge Calculator
Simulation
Coming soon...
