Titrations
Core Concept
A titration is a laboratory method used to determine the concentration of an unknown solution by reacting it with a solution of known concentration (the titrant).
Key Principle: The reaction between the titrant and analyte (unknown solution) must be stoichiometrically balanced.
Practice Tips
Titrations are precise and widely used techniques to determine unknown concentrations.
In acid-base titrations, choose an appropriate indicator and understand the reaction's equivalence point.
Use M1V1=M2V2M_1V_1 = M_2V_2M1V1=M2V2 for titration calculations and always verify your results.
Analyze titration curves to understand the pH changes during the process.
Podcast Episode
Types of Titrations
Acid-Base Titrations
Process: Involve the neutralization reaction between an acid and a base.
Example: Determining the concentration of $HCl$ using $NaOH$ as the titrant.
Redox Titrations
Process: Based on oxidation-reduction reactions where electrons are transferred between the analyte and the titrant.
Example: Determining the concentration of $Fe^{2+}$ using $KMnO_4$ (Potassium Permanganate).
Complexometric Titrations
Process: Involve the formation of a stable complex ion.
Example: Determining metal ions (like $Ca^{2+}$ or $Mg^{2+}$) using EDTA as the complexing agent.
Precipitation Titrations
Process: Based on a reaction that forms an insoluble precipitate.
Example: Determining $Cl^-$ concentration using $AgNO_3$ (Argentometric titration), which forms a white $AgCl$ precipitate.
Steps in a Titration
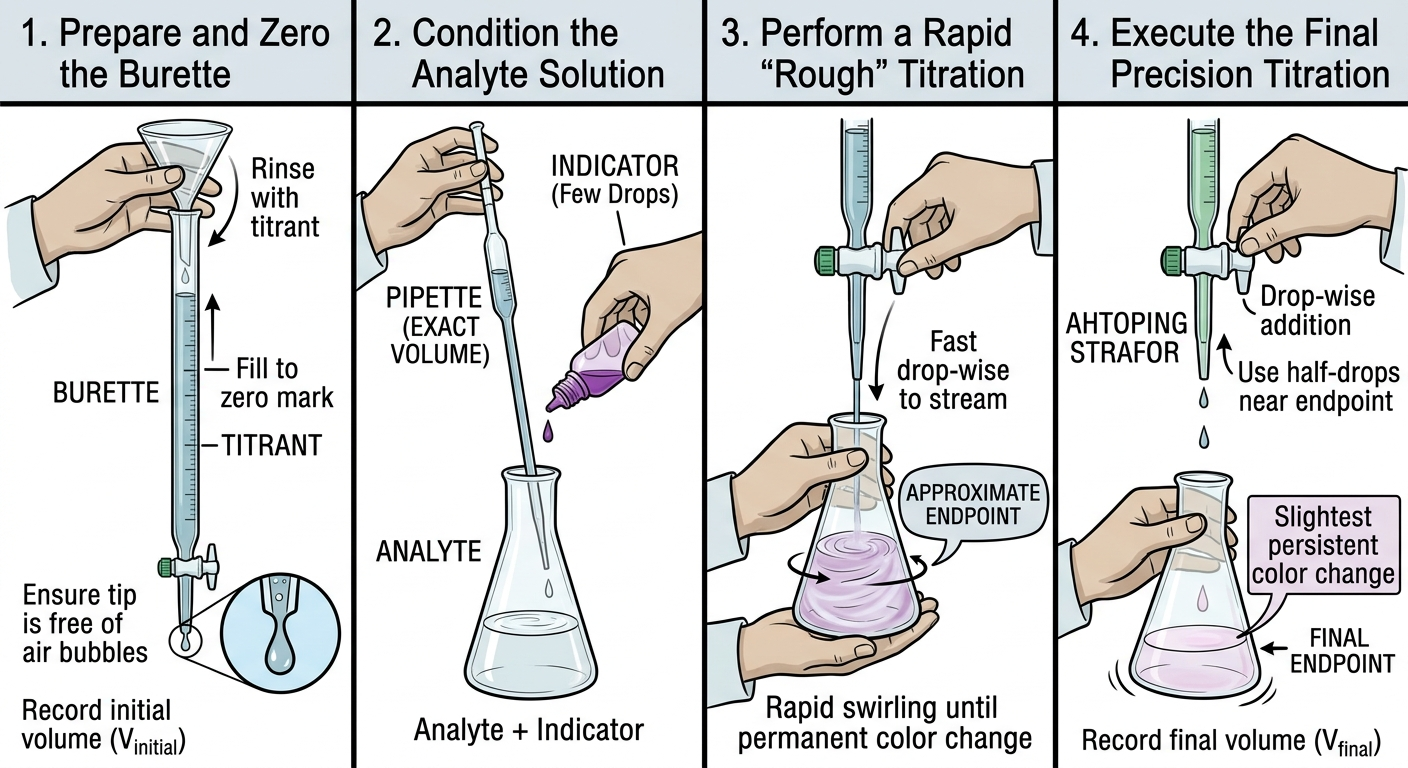
Acid-Base Titrations
Neutralization Reaction
General Reaction: Acid + Base → Salt + Water
Example:
$$HCl + NaOH \rightarrow NaCl + H_2O$$
Key Terms
Equivalence Point: The point at which the moles of acid equal the moles of base. For strong acid-strong base titrations, this occurs at $pH = 7$.
End Point: The point where the indicator changes color, ideally very close to the equivalence point.
Indicators: Substances chosen to change color at the specific pH of the equivalence point.
Phenolphthalein: Colorless in acid, pink in base ($pH$ range 8.2–10).
Methyl Orange: Red in acid, yellow in base ($pH$ range 3.1–4.4).
Titration Curve
A titration curve plots the pH of the solution against the volume of titrant added.
Strong Acid-Strong Base:
Starts at a low pH (acidic).
Sharp increase at the equivalence point ($pH = 7$).
Levels off at a high pH (basic).
Weak Acid-Strong Base:
Starts at a slightly higher pH.
Equivalence point above $pH = 7$ due to the formation of a weak conjugate base.
Weak Base-Strong Acid:
Starts at a slightly lower pH.
Equivalence point below $pH = 7$ due to the formation of a weak conjugate acid.
Titration Calculations
Key Formula: $$M_1V_1 = M_2V_2$$
Where:
$M_1$: Molarity of the titrant
$V_1$: Volume of the titrant
$M_2$: Molarity of the analyte
$V_2$: Volume of the analyte
